
Here is the list of electron configurations of the first 20 elements of periodic table. Electron configuration of elements (List) The crystal structure of ruthenium is HCP (Hexagonal close packing). The melting point of ruthenium is 2334 ☌ and its boiling point is 4150 ☌. The atomic mass of ruthenium is 101.07 u and its density is 12.37 g/cm 3. The shells and subshells of an atom determine its electron configuration, which in turn determines its chemical and physical properties. Ruthenium is a solid metal having Silvery white metallic lustre. 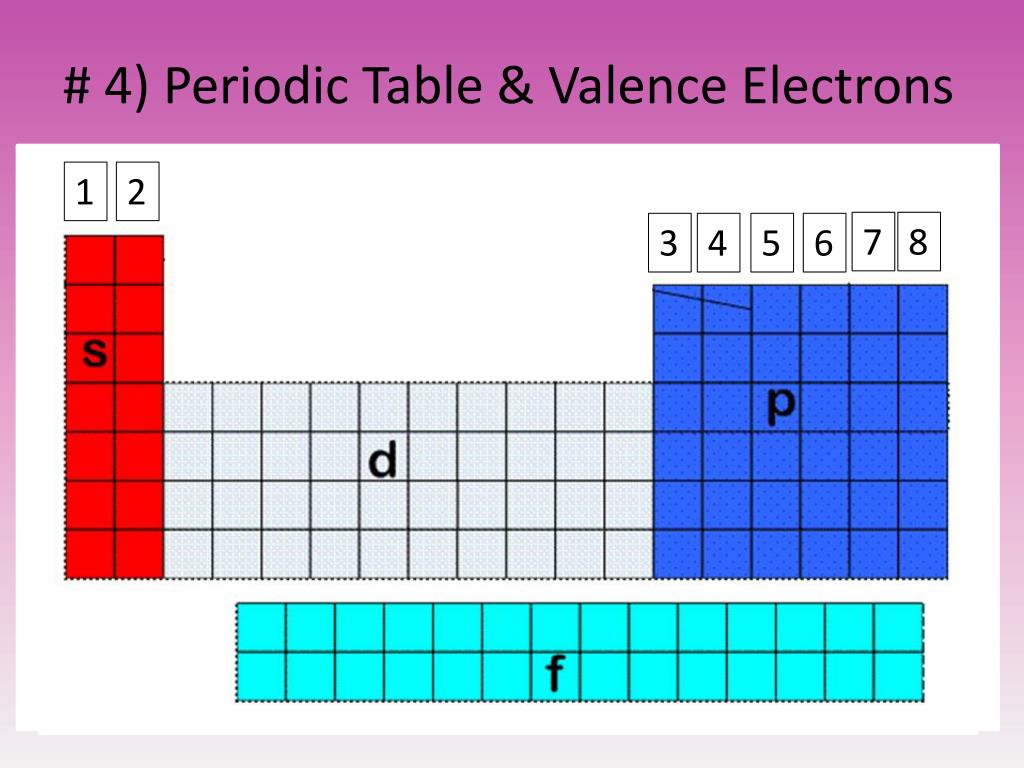 
The s subshell can hold a maximum of 2 electrons, the p subshell can hold up to 6 electrons, the d subshell can hold up to 10 electrons, and the f subshell can hold up to 14 electrons. Subshell (or Orbitals): Within each shell, there are different subshells, which are labeled using letters such as s, p, d, and f. The first shell can only hold a maximum of 2 electrons, while the second shell can hold up to 8 electrons, and subsequent shells can hold even more. Shell: The shell is the orbit in which the electrons revolve around the nucleus of an atom.Įach shell is assigned a number (n), starting from one for the first shell and increasing with each additional shell. What are Shells (n) and Subshells (s, p, d, f)? The arrangement of electrons in an atom is determined by its energy levels (which are also called shells), and the specific subshells within those shells.Įlectron configuration is typically represented using a notation that indicates the number of electrons in each shell and subshell, and it follows certain rules based on the principles of quantum mechanics. The electron configuration of an atom determines its chemical and physical properties, such as its reactivity, conductivity, and melting point. Electronic configuration refers to the arrangement of electrons in an atom.Įlectrons are tiny negatively charged particles that orbit the positively charged nucleus of an atom.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
